Choosing suitable parts for medical device repair is crucial for maintaining device functionality and safety. The medical device industry demands high standards. Any part used must meet precise specifications. This ensures patient safety and device reliability.
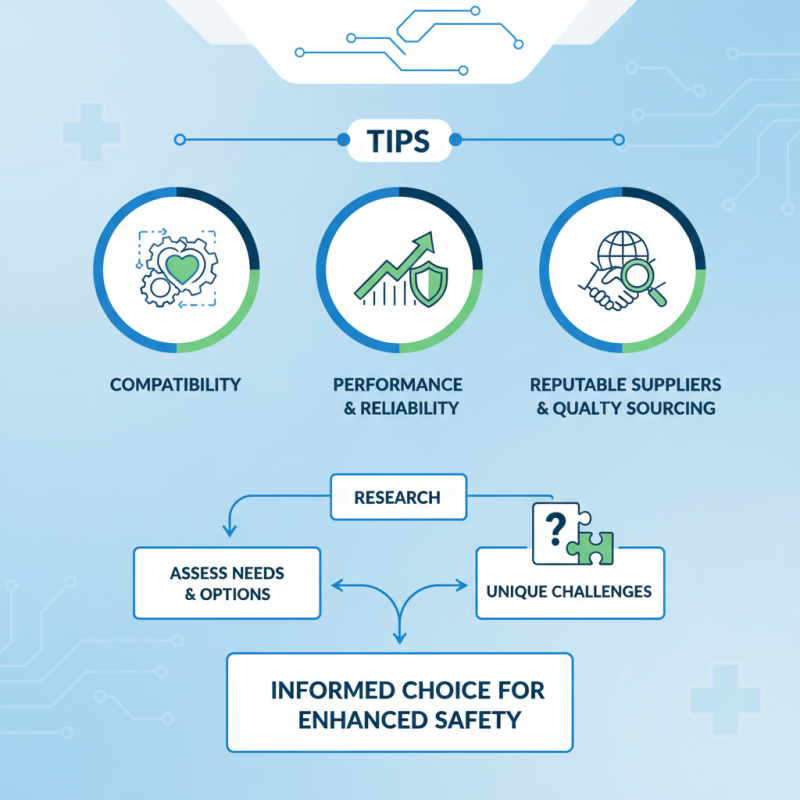
When discussing how to choose suitable parts for medical device repair, one must consider various factors. Compatibility with the existing system is vital. Components should also have a track record of performance. Sourcing from reputable suppliers adds to this reliability. Quality should never be compromised.
However, it is not always straightforward. With so many options available, the decision can be overwhelming. Each repair situation may reveal unique challenges. It is essential to do thorough research before making a final choice. Understanding these nuances can lead to better repair outcomes and enhanced safety.
Selecting parts for medical device repair requires a deep understanding of regulatory compliance. Compliance ensures that any components used meet safety and efficacy standards. The FDA has strict guidelines which all medical device manufacturers must adhere to. In fact, the FDA reports that around 3,000 recalls occur annually, often due to non-compliance in parts selection. This highlights the critical importance of choosing the right components.
When selecting parts, always consult industry standards. Look for components that comply with ISO 13485. This standard focuses on quality management systems for medical devices. Additionally, consider the biocompatibility of materials. The ISO 10993 standard outlines testing methods for assessing materials' safety in medical applications. Regular audits and checks can help ensure compliance over time.
Here are some tips: Review any past product recalls related to similar devices. Keep up to date with regulatory changes. Know that "off-the-shelf" parts might not meet specific device needs. Always document the selection process thoroughly. This will not only ensure compliance but also improve the reliability of repairs. Remember, regulatory compliance is not just a checkbox—it's crucial for patient safety.
| Criteria | Description | Regulatory Compliance | Example Parts |
|---|---|---|---|
| Material Compatibility | Ensure materials are suitable for medical use and do not cause adverse reactions. | FDA 21 CFR Part 820 | Silicone, Polycarbonate |
| Sterilization Methods | Select parts that can withstand approved sterilization processes. | ISO 17665 | Surgical Instruments |
| Documentation and Traceability | Ensure all parts are documented for traceability through the supply chain. | FDA 21 CFR Part 820.30 | Compliance Certificates |
| Performance Standards | Select parts that meet the required performance specifications for safety and efficacy. | IEC 60601 | Electro-medical Devices |
| Supplier Certification | Ensure suppliers are certified and comply with Good Manufacturing Practices (GMP). | ISO 13485 | OEM Components |
When selecting parts for medical device repair, understanding quality standards is vital. ISO 13485, a global standard, focuses on quality management systems. It ensures that medical devices consistently meet regulatory requirements. Compliance with these standards aids in enhancing safety and effectiveness. Each part's quality influences the overall device performance.
Tips for selecting parts include verifying documentation. Always check for relevant certifications. This ensures the parts meet the necessary regulations. Don’t hesitate to ask for test reports. Another essential step is to evaluate suppliers. Their reputation impacts the quality of parts. Research their history of compliance and reliability.
FDA guidelines also play a significant role. They provide a framework for ensuring safety and efficacy. Adhering to these guidelines can prevent costly recalls. Engage in thorough due diligence before finalizing suppliers. Reflect on past repair experiences to guide future selections. The journey to quality parts often requires ongoing evaluation and improvement.
Selecting the right parts for medical device repair is critical. Assessing material properties plays a significant role in this process. The compatibility of materials affects the functionality and safety of the device. Each component must match the specifications of the original design. This ensures reliability and adherence to industry standards.
Consider the mechanical properties of materials. For instance, stiffness, tensile strength, and fatigue resistance are essential in maintaining the device's integrity. Additionally, biocompatibility is crucial. Devices often interact with biological tissues, so materials need to be safe and non-toxic. This can sometimes complicate repair efforts. It might not always be easy to find matching materials that meet both mechanical and biological criteria.
It's also important to evaluate the materials' long-term performance. Some parts may degrade faster than anticipated. Understanding factors like environmental exposure and wear can guide repair decisions. It’s a continuous learning process. One must reflect on past repair attempts. Were the right materials chosen? Did they perform as expected? These reflections can lead to better decisions in future repairs.
When selecting parts for medical device repair, supplier credentials are vital. Companies must prioritize certifications in relevant manufacturing standards. These credentials ensure the supplier adheres to strict quality control measures. A reliable supplier often displays industry recognition, enhancing their credibility.
Track records are equally important. Investigating a supplier's history provides insight into their reliability. Consider their past performance with other medical devices. Look for reviews and testimonials from previous clients. This feedback can reveal potential red flags. A supplier with a history of on-time deliveries and high customer satisfaction scores can be a strong candidate.
However, not every supplier meets expectations. Some might boast impressive credentials, yet fail in execution. Delayed shipments or poor product quality can hinder device repairs. It's essential to remain critical and conduct thorough due diligence. A proactive approach in evaluating suppliers will better protect patient safety and operational efficiency.
When repairing medical devices, cost-effectiveness plays a crucial role. Balancing quality and expense is essential for providing reliable solutions. Often, professionals face pressure to cut costs, which can lead to potential risks. Selecting the right parts while ensuring safety is paramount.
One key tip is to assess the part's performance history. Look for components with proven reliability. Conduct thorough research on alternatives that still meet necessary standards. Lower-priced parts can sometimes underperform or fail sooner than expected. Weigh the risks of possible downtime against the initial savings.
Another consideration is the supplier's reputation. Partners with a solid track record of quality assurance can help maintain standards. Communicate openly with suppliers about your needs. Evaluate their response and support during the selection process. They should provide reliable assistance and documentation. Investing extra time in this step could save significant costs in the long run.
: Understanding quality standards ensures medical devices meet regulatory requirements. This enhances safety and effectiveness.
ISO 13485 is a global standard for quality management systems in the medical field. It promotes consistent regulatory compliance.
Always check for relevant certifications and ask for test reports. This confirms parts meet necessary regulations.
A supplier's reputation affects part quality. Research their compliance history for reliable choices.
FDA guidelines provide safety and efficacy frameworks. Following them can prevent costly product recalls.
Ensure materials match the original specifications. This guarantees functionality and maintains device safety.
Stiffness, tensile strength, and fatigue resistance are vital for device integrity. Evaluate these factors carefully.
Analyze external factors like wear and environmental exposure. Reflect on past repairs to improve future decisions.
Lower-priced components may underperform or fail sooner. This could lead to unexpected costs and downtime.
Open communication helps clarify your needs and ensures reliable support. This can improve part selection and performance.
When considering how to choose suitable parts for medical device repair, it is crucial to understand the regulatory landscape surrounding medical devices. Ensuring compliance with regulations, such as ISO 13485 and FDA guidelines, is foundational to any parts selection process. Quality standards must be evaluated to guarantee that the components used in repairs meet the necessary safety and effectiveness criteria.
Another significant factor is assessing compatibility, which involves understanding material properties that impact the device's performance post-repair. Additionally, analyzing supplier credentials—such as certifications and a proven track record—adds another layer of assurance regarding the quality and reliability of the parts. Finally, implementing a cost-effective strategy that balances quality and expense will help in making informed decisions while conducting repairs. In summary, a thorough evaluation across these dimensions will facilitate the selection of appropriate parts for successful medical device repair.

 Xinli Technology
Xinli Technology